See how to use this calculator for step-by-step guide. 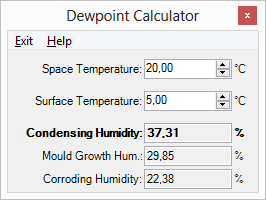
Introduction to Chemical Engineering Thermodynamics (8th ed.). Bear in mind that flash calculation can't be calculated ifįeed exists as subcooled liquid or superheated vapor. Where \(z_i\) is molar composition of the inlet feed, \(V\) is the vapor fractions, and \(L\) is the liquid fractions or \(L=1-V\). While for dew point calculations, the unknown liquid-phase compositions can be simplified by asumming \(\sum x_i=1\), so thatįor flash calculation, we calculate the composition based on material balance of the system, 
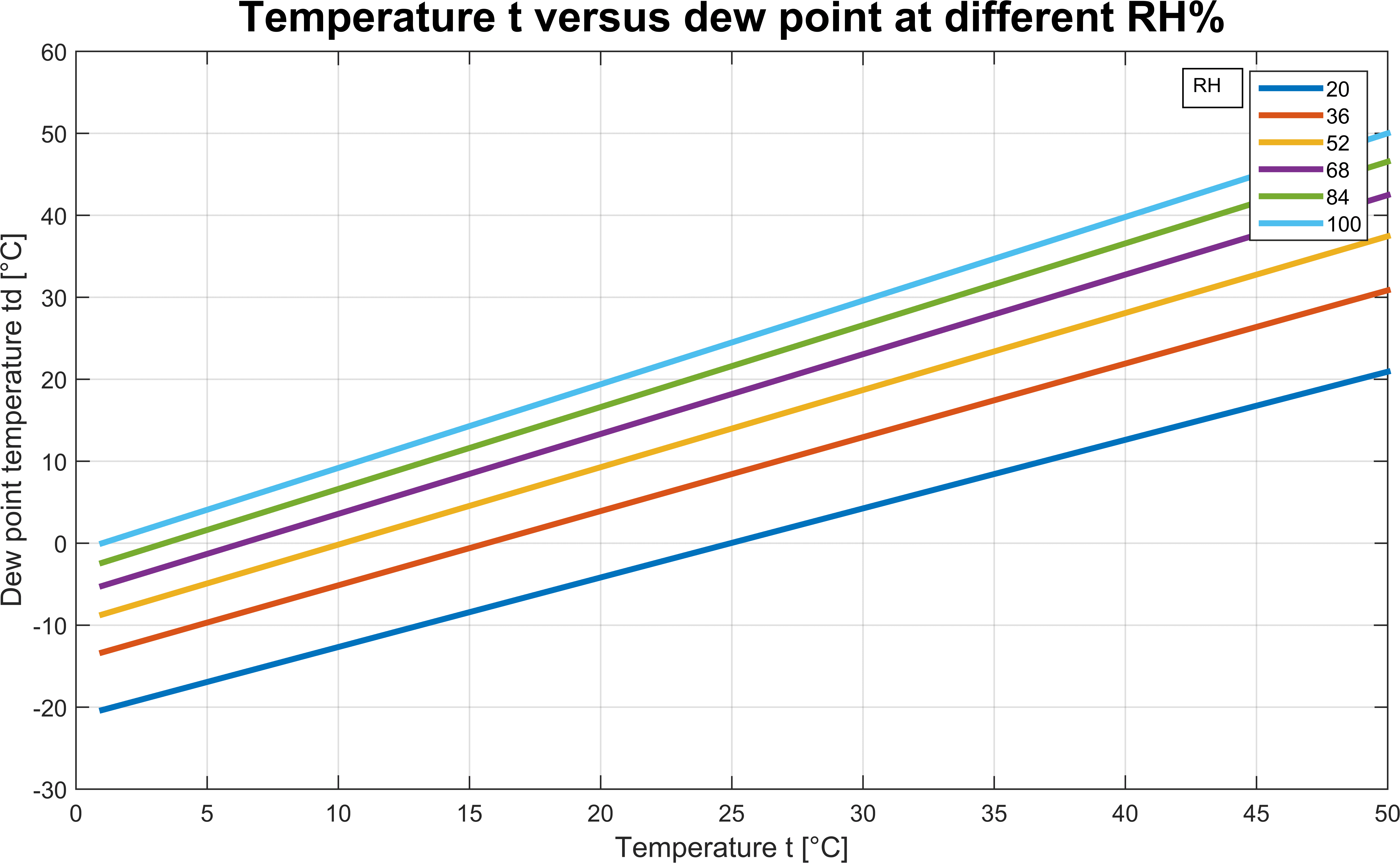
Likewise, given the frost point Tf, we can calculate the relative humidity. P_i^, given \(P\) and \(T\)įor bubble point calculations which vapor-phase compositions are unknown, we can simplify the calculations by asumming \(\sum y_i=1\), so that the Raoult's law become (1) gives RHl as an explicit function of temperature T and dewpoint Td. The mathematical equations which reflects the Raoult's law based on the two assumptions above is The liquid phase is an ideal solution, which means it only valid if the chemical species comprise are chemically similar.The vapor phase is an ideal gas, which means it can apply only from low to moderate pressures, and.There are two major assumptions in Raoult's law, which are: Raoult's law states that the vapor pressure of a solvent above a solution is equal to the vapor pressure of the pure solvent at the same temperature scaled by the mole fraction of the solvent present. To perform bubble and dew point calculation, first we must understand the theory of Raoult's Law for ideal mixture. Bubble Point, Dew Point, and Flash Calculation
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
